 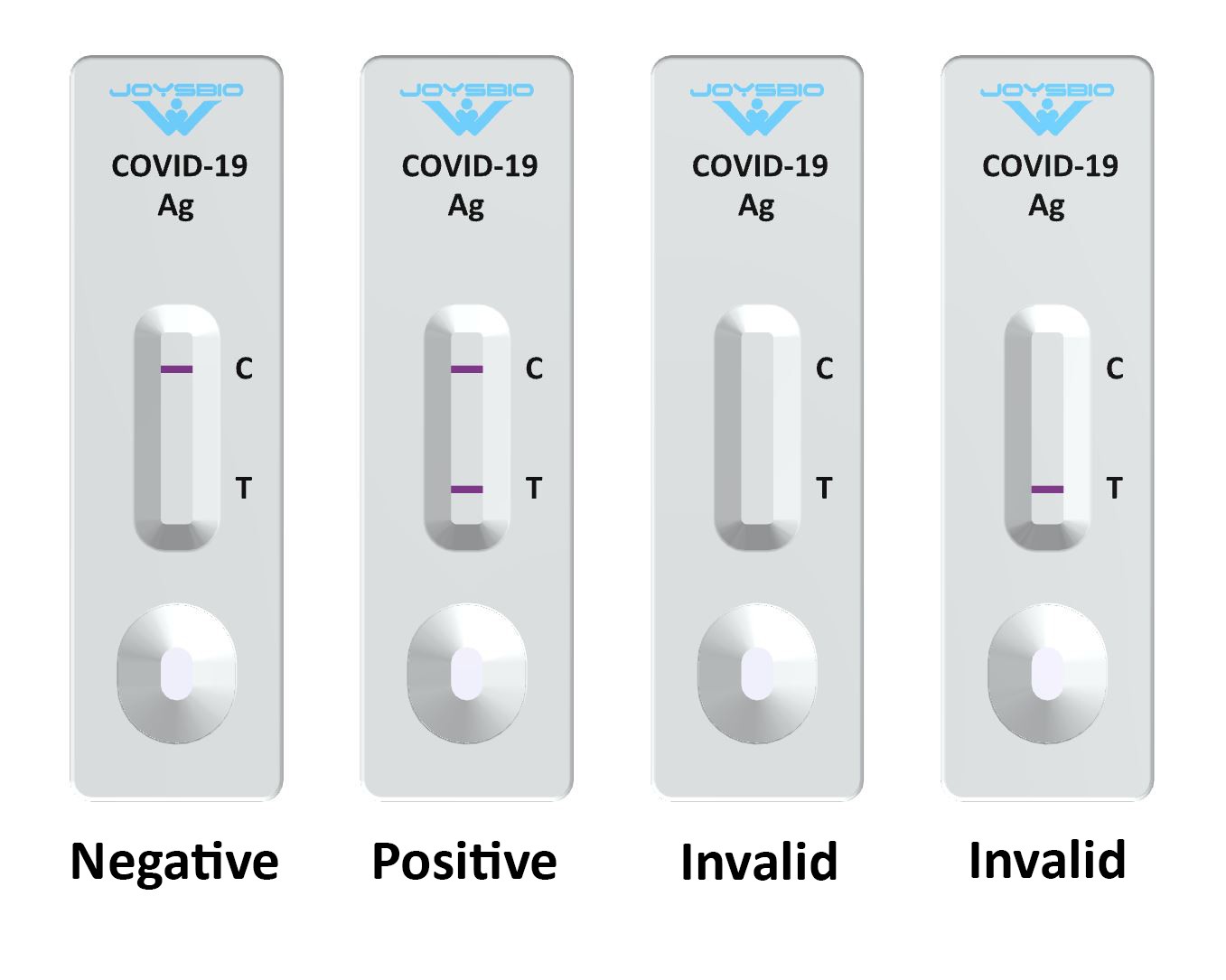
Various studies are done to evaluate the diagnostic accuracy of Ag-RDTs that vary in study design, study setting and study population along with various brands being evaluated. This trade-off for the ease-of-use of Ag-RDTs is decrease in sensitivity and specificity of test. Antigen detection rapid diagnostic tests (Ag-RDTs) used for detecting SARS-CoV-2 nucleocapsid protein are inexpensive, faster and easy to use alternative of NAAT for COVID-19 testing. 
Although, NAAT is the preferred initial diagnostic test for COVID-19, its use in low resource settings is limited by its cost and need for qualified clinical laboratory personnel. CDC has outlined viral test (Nucleic Acid Amplification Test (NAAT) and antigen tests) and antibody tests as methods for testing current and past infection with COVID-19 respectively. In addition to strategies like vaccination against COVID-19, contact tracing, and home isolation for potential exposure to or diagnosis of COVID-19, early diagnosis and community screening for COVID-19 are crucial to control the spread of disease. However, false negative results need to be interpreted with caution.Ĭoronavirus disease 2019 (COVID-19) has been spreading rapidly across the globe causing loss of millions of lives since it was first reported in Wuhan, China and later declared a pandemic on 11 th March, 2020. Ag-RDTs, therefore have the potential to be used as a screening tool for SARS-CoV-2 detection in low resource settings where RT-PCR might not be readily accessible. The review demonstrated pooled sensitivity value approaching the minimum performance requirement for diagnosis of COVID-19 by WHO with specificity value meeting the specified requirement. However, an inverse relation between cycle threshold (Ct) and sensitivity of Ag-RDT was not seen. Sensitivity was marginally higher in subgroup analysis based on studies with low risk of bias and applicability concerns (78.9%) and studies using SD Biosensor Ag-RDT (79.4%). From the total of 12 diagnostic accuracy studies with 4,817 study participants, pooled sensitivity and specificity were 78.2% and 99.5% respectively. The analysis was done using Review Manager 5.4 and R software 4.0.2. The Quality Assessment of Diagnostic Accuracy Studies (QUADAS)-2 tool was used to assess the methodological quality of studies. A systematic search was conducted in PubMed, CINAHL, Embase, Scopus, and Google Scholar to identify studies published between 1 January, 2020 and 15 August, 2021. We included peer reviewed prospective or retrospective cohort studies, cross-sectional studies, case control studies, randomized clinical trials (RCTs) as well as non-randomized experimental studies which addressed the review question. The study population comprised of people living in LMICs irrespective of age and gender, who had undergone testing for COVID-19. We included studies that evaluated the diagnostic accuracy of Ag-RDTs (sensitivity and specificity) against reverse transcription-polymerase chain reaction (RT-PCR) as a reference standard. In this systematic review and meta-analysis, we assessed the diagnostic accuracy of Ag-RDTs in low and middle-income countries (LMICs). Antigen detection rapid diagnostic tests (Ag-RDTs) used for detecting severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) nucleocapsid protein are inexpensive, faster and easy to use alternative of Nucleic Acid Amplification Test (NAAT) for diagnosis of Coronavirus disease 2019 (COVID-19). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
